Preclinical and Experimental Surgery Solutions
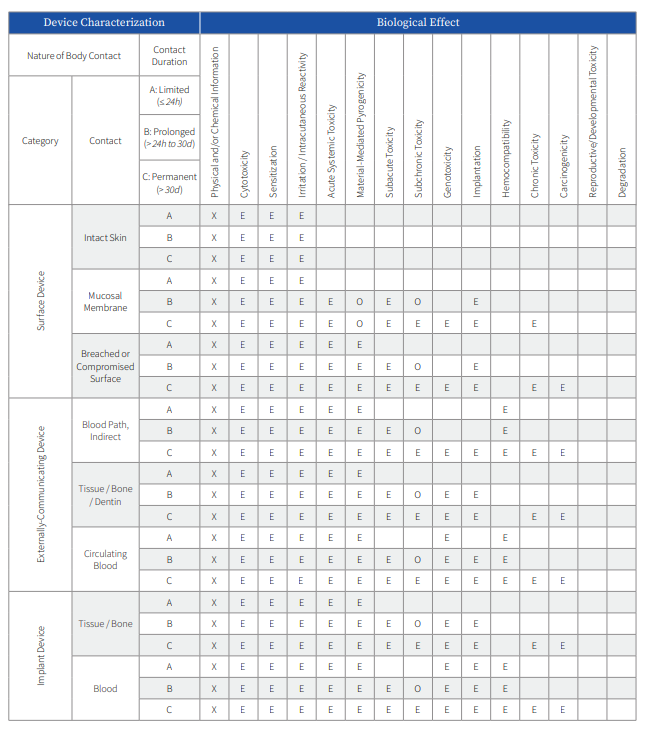
Biocompatibility endpoints required for assessment as a part of a risk based evaluation of medical device safety are provided by ISO 10993-1 and various international regulatory body standards and guidance documents. The endpoints detailed in the matrix table describe the potential risks based on the clinical contact type and duration of the medical device to the patient or clinician. Devices may fall into multiple categories based upon the various components (delivery system and implant), and it is important to consult all available applicable standards and guidance documents which may provide other specifics to the medical device biocompatibility evaluation process. Each endpoint for evaluation should be identified as a part of a biological evaluation plan (BEP).